

Journal of Clinical and Biomedical Sciences
DOI: 10.58739/jcbs/v16i1.24.204
Year: 2026, Volume: 16, Issue: 1, Pages: 63-68
Original Article
Santosh Kumar1, Jasmeen Syan2*, Ankit Kumar2
1School of Pharmacy, Maya Devi University, Dehradun, Uttarakhand, India.
2College of Pharmacy, Shivalik Campus, Dehradun, Uttarakhand, India
*Corresponding Author
Email: [email protected]
Received Date:04 November 2024, Accepted Date:29 December 2025, Published Date:20 April 2026
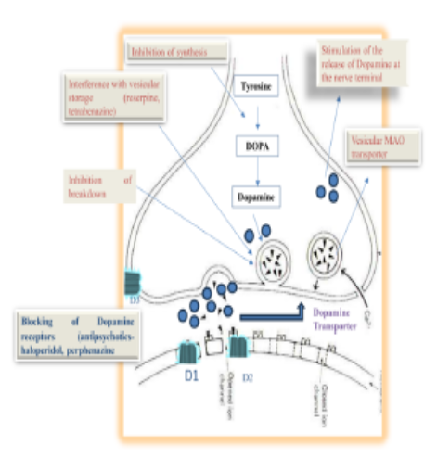
Mental illness is a critical aspect of human health that significantly affects individuals' emotional and psychological well-being. The present study focused on the “Current status of mental health which highlights both advancements and persistent challenges.” About 10% of people worldwide suffer from psychosis, a mental illness characterized by severe distortions in cognition, behavior, perception (delusions and hallucinations), and the ability to identify reality. The growing interest in natural remedies for mental health issues in recent years is indicative of a move toward more comprehensive methods of treating ailments including stress, anxiety, and depression. Numerous genetic, nutritional, microbial, and environmental variables are involved in this complex illness. However, several conditions are included under psychotic illnesses. Although they are pharmacologically categorized as dopamine receptor antagonists, many of them also act on other targets, such as 5-hydroxytryptamine (5-HT) receptors, which may contribute to some of their therapeutic efficacy. The activation of the immune system, neuro-inflammation, endocannabinoid receptors, neurotransmission, cell signaling pathways, and oxidative stress status are all changed and modified by these phytochemicals. Most phytochemicals with anti-schizophrenic properties include flavonoids, alkaloids, terpenoids, polypropanoids, lactones, and glycosides.
Keywords: Psychosis, Phytochemicals, Medications, Receptors, Schizophrenia, Disease, Anti-psychotics
1. Calabrese J, Al Khalili Y, Shaheen K. Psychosis (nursing). 2023 May. StatPearls Publishing.
2. Adityanjee, Aderibigbe YA, Theodoridis D, Vieweg WV. Dementia praecox to schizophrenia: The first 100 years. Psychiatry and Clinical Neurosciences. 1999; 53 (4). Available from: https://doi.org/10.1046/j.1440-1819.1999.00584.x
3. Orsolini L, Pompili S, Volpe U. Schizophrenia: A Narrative Review of Etiopathogenetic, Diagnostic and Treatment Aspects. Journal of Clinical Medicine. 2022; 11 (17). Available from: https://doi.org/10.3390/jcm11175040
4. Blakemore SJ, Frith CD. Functional Neuroimaging Studies of Schizophrenia. Brain Mapping: The Disorders. 2000; Available from: https://doi.org/10.1016/b978-012481460-8/50024-x
5. Brown AS, Derkits EJ. Prenatal Infection and Schizophrenia: A Review of Epidemiologic and Translational Studies. American Journal of Psychiatry. 2010; 167 (3). Available from: https://doi.org/10.1176/appi.ajp.2009.09030361
6. Cannon TD. Neurodevelopmental influences in the genesis and epigenesis of schizophrenia: An overview. Applied and Preventive Psychology. 1998; 7 (1). Available from: https://doi.org/10.1016/s0962-1849(98)80021-8
7. Boksa P. Abnormal synaptic pruning in schizophrenia: Urban myth or reality?. Journal of Psychiatry and Neuroscience. 2012; 37 (2). Available from: https://doi.org/10.1503/jpn.120007
8. Woods BT. Is Schizophrenia a Progressive Neurodevelopmental Disorder? Toward a Unitary Pathogenetic Mechanism. American Journal of Psychiatry. 1998; 155 (12). Available from: https://doi.org/10.1176/ajp.155.12.1661
9. Jun R, Zhang W, Beacher NJ, Zhang Y, Li Y, Lin DT. Dysbindin-1, BDNF, and GABAergic Transmission in Schizophrenia. Frontiers in Psychiatry. 2022; 13 Available from: https://doi.org/10.3389/fpsyt.2022.876749
10. Gejman PV, Sanders AR, Duan J. The Role of Genetics in the Etiology of Schizophrenia. Psychiatric Clinics of North America. 2010; 33 (1). Available from: https://doi.org/10.1016/j.psc.2009.12.003
11. Mei L, Xiong WC. Neuregulin 1 in neural development, synaptic plasticity and schizophrenia. Nature Reviews Neuroscience. 2008; 9 (6). Available from: https://doi.org/10.1038/nrn2392
12. Brown AS. The environment and susceptibility to schizophrenia. Progress in Neurobiology. 2011; 93 (1). Available from: https://doi.org/10.1016/j.pneurobio.2010.09.003
13. Singh S, Khanna D, Kalra S. Role of Neurochemicals in Schizophrenia. Current Psychopharmacology. 2020; 9 (2). Available from: https://doi.org/10.2174/2211556009666200401150756
14. Reynolds GP. Beyond the Dopamine Hypothesis. British Journal of Psychiatry. 1989; 155 (3). Available from: https://doi.org/10.1192/bjp.155.3.305
15. Meltzer HY, Stahl SM. The Dopamine Hypothesis of Schizophrenia: A Review*. Schizophrenia Bulletin. 1976; 2 (1). Available from: https://doi.org/10.1093/schbul/2.1.19
16. Seeman P. Dopamine receptors and the dopamine hypothesis of schizophrenia. Synapse. 1987; 1 (2). Available from: https://doi.org/10.1002/syn.890010203
17. Coyle JT. NMDA Receptor and Schizophrenia: A Brief History. Schizophrenia Bulletin. 2012; 38 (5). Available from: https://doi.org/10.1093/schbul/sbs076
18. McCutcheon RA, Krystal JH, Howes OD. Dopamine and glutamate in schizophrenia: biology, symptoms and treatment. World Psychiatry. 2020; 19 (1). Available from: https://doi.org/10.1002/wps.20693
19. Van Berckel BN. Glutamate and Schizophrenia. Current Neuropharmacology. 2003; 1 (4). Available from: https://doi.org/10.2174/1570159033477026
20. Gardner DM, Baldessarini RJ, Waraich P. Modern antipsychotic drugs: a critical overview. Canadian Medical Association Journal. 2005; 172 (13). Available from: https://doi.org/10.1503/cmaj.1041064
21. Wetchateng T, Piyabhan P. EPA-0117 – The use of bacosides a and b to prevent a cognitive deficit in schizophrenia rat models resulting in increased vesicular glutamate transporter 2 (VGLUT2) in the cingulate gyrus. European Psychiatry. 2014; 29 Available from: https://doi.org/10.1016/s0924-9338(14)77591-1
22. Shadkami F, Jones ADs. Nontargeted Profiling of Specialized Metabolites ofDigitalis purpureawith a Focus on Cardiac Glycosides. ACS Symposium Series. 2012; Available from: https://doi.org/10.1021/bk-2012-1093.ch011
23. Yadav M, Jindal DK, Dhingra MS, Kumar A, Parle M, Dhingra S. Protective effect of gallic acid in experimental model of ketamine-induced psychosis: possible behaviour, biochemical, neurochemical and cellular alterations. Inflammopharmacology. 2018; 26 (2). Available from: https://doi.org/10.1007/s10787-017-0366-8
24. Jie F, Yang X, Wu L, Wang M, Lu B. Linking phytosterols and oxyphytosterols from food to brain health: origins, effects, and underlying mechanisms. Critical Reviews in Food Science and Nutrition. 2022; 62 (13). Available from: https://doi.org/10.1080/10408398.2020.1867819
25. Kucerova J, Tabiova K, Drago F, Micale V. Therapeutic Potential of Cannabinoids in Schizophrenia. Recent Patents on CNS Drug Discovery. 2014; 9 (1). Available from: https://doi.org/10.2174/1574889809666140307115532
26. Georgiadou G, Grivas V, Tarantilis PA, Pitsikas N. Crocins, the active constituents of Crocus Sativus L., counteracted ketamine–induced behavioural deficits in rats. Psychopharmacology. 2014; 231 (4). Available from: https://doi.org/10.1007/s00213-013-3293-4
27. Sun X-J, Zhao X, Xie J-N, Wan H. Crocin alleviates schizophrenia-like symptoms in rats by upregulating silent information regulator-1 and brain derived neurotrophic factor. Comprehensive Psychiatry. 2020; 103 Available from: https://doi.org/10.1016/j.comppsych.2020.152209
28. Heresco-Levy U, Ermilov M, Lichtenberg P, Bar G, Javitt DC. High-dose glycine added to olanzapine and risperidone for the treatment of schizophrenia. Biological Psychiatry. 2004; 55 (2). Available from: https://doi.org/10.1016/s0006-3223(03)00707-8
29. Leiderman E, Zylberman I, Zukin SR, Cooper TB, Javitt DC. Preliminary investigation of high-dose oral glycine on serum levels and negative symptoms in schizophrenia: an open-label trial. Biological Psychiatry. 1996; 39 (3). Available from: https://doi.org/10.1016/0006-3223(95)00585-4
30. Hudepohl NS, Nasrallah HA. Antipsychotic drugs. Handbook of Clinical Neurology. 2012; Available from: https://doi.org/10.1016/b978-0-444-52002-9.00039-5
31. Miyamoto S, Merrill DB, Fredrik Jarskog L, Wolfgang Fleishhacker W, Marder SR, Lieberman JA. Antipsychotic Drugs. Psychiatry. 2015; Available from: https://doi.org/10.1002/9781118753378.ch104
32. Strange PG. Antipsychotic Drugs: Importance of Dopamine Receptors for Mechanisms of Therapeutic Actions and Side Effects. Pharmacological Reviews. 2001; 53 (1). Available from: https://doi.org/10.1016/s0031-6997(24)01483-2
33. Palermo-Neto J. Dopaminergic systems: dopamine receptors. Psychiatric Clinics of North America. 1997; 20 (4). Available from: https://doi.org/10.1016/s0193-953x(05)70341-5
34. Schaus JM, Clemens JA. Chapter 5. Dopamine Receptors and Dopaminergic Agents. Annual Reports in Medicinal Chemistry. 1985; Available from: https://doi.org/10.1016/s0065-7743(08)61031-3
35. Meltzer HY. The Mechanism of Action of Novel Antipsychotic Drugs. Schizophrenia Bulletin. 1991; 17 (2). Available from: https://doi.org/10.1093/schbul/17.2.263
36. Davis JM, Casper R. Antipsychotic Drugs. Drugs. 1977; 14 (4). Available from: https://doi.org/10.2165/00003495-197714040-00002
37. Porsolt RD, Moser PC, Castagné V. Behavioral Indices in Antipsychotic Drug Discovery. The Journal of Pharmacology and Experimental Therapeutics. 2010; 333 (3). Available from: https://doi.org/10.1124/jpet.110.166710
38. Rang HP, Dale MM, Ritter JM, Flower RJ. Rang and Dale’s Pharmacology. 6th edition Churchill Livingstone Publishers. 2007; Available from: https://doi.org/10.1016/b978-0-443-06911-6.50043-8
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Published By Sri Devaraj Urs Academy of Higher Education, Kolar, Karnataka
Subscribe now for latest articles and news.